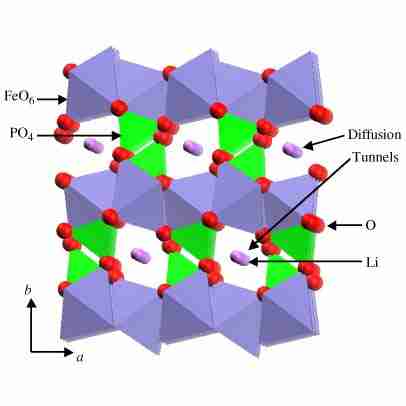
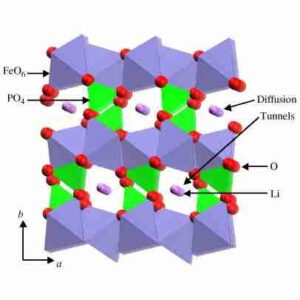
Lithium iron phosphate (LiFePO4), also called LFP, is one of the more recently developed rechargeable battery cathodes and is a variation of lithium-ion chemistry. Rechargeable lithium iron phosphate batteries use LiFePO4 as the cathode material and graphitic carbon as the anode. Despite having a lower energy density than other lithium-ion chemistries, lithium-iron phosphate batteries provide better power density and longer life cycles. The LiFePO4 powder is usually carbon-coated to improve its conductivity for its use in batteries. Carbon-coated lithium iron phosphate (C-LiFePO4) powders have been produced commercially on a large scale with a controlled solid-state reaction.
Specifications
| Item | Value |
|---|---|
| D50 (um) | 1.302 |
| BET (m2/g) | 13.36 |
| Tap Density (g/cm3) | 1.14 |
| Discharge capacity in half cell (mAh/g) | 153 |
| Test condition | 25 degree, 0.1C rate, 2.0~4.2V |
Note:
- As the primary particles of the Lithium iron phosphate (LiFePO4) are nano-sized, and the specific surface area is large, more binder is necessary for the electrode formula.
- Carbon-coated aluminum foil is suggested as the current collector. This is to improve the contact between the active material and substrate and thus reduce the internal resistance of the battery. It can improve the adhesion of the electrode to the substrate.
- The tap density of the LiFePO4 electrode is suggested to be ~2.0g/cc.




Reviews
There are no reviews yet.